Philips CPAP & BiPAP Lawsuit Attorneys
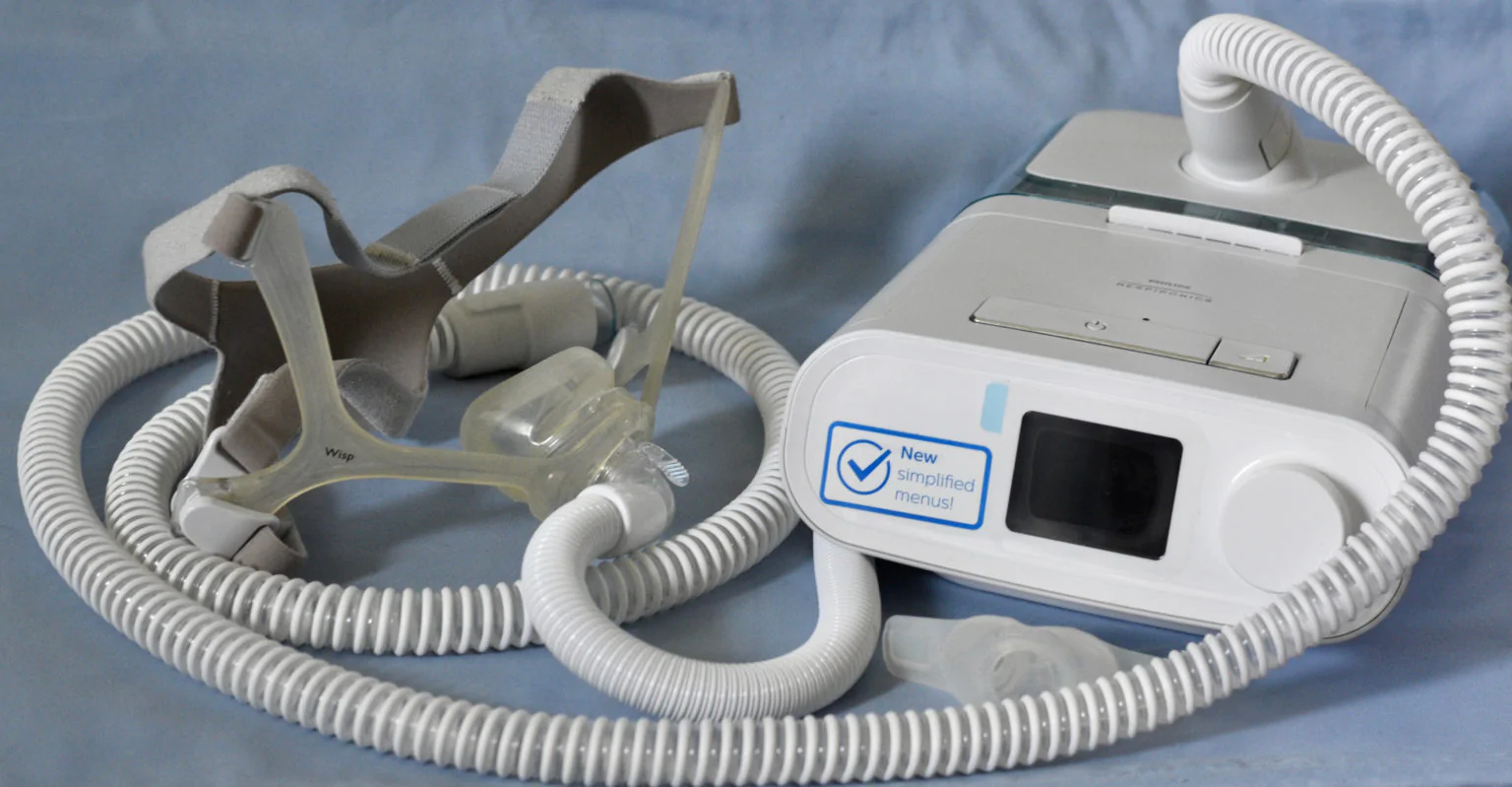
Philips Respironics has issued a recall for their continuous positive airway pressure devices (CPAP) and BiLevel positive airway pressure (BiPAP) devices. Recent studies revealed these machines, used to treat sleep apnea, contain foam that may degrade over time and become toxic, putting users at risk for several types of cancers and long-term health issues.
Customers that have been using a Philips device and are experiencing symptoms of illness may be eligible to file a toxic tort lawsuit. This gives victims the chance to receive compensation for any related medical expenses, lost wages, and other damages such as pain and suffering—and helps hold Philips responsible for selling harmful products.
Cancers & Short-Term Risks Likely Caused by CPAP and BiPAP Devices
The toxic materials that users are exposed to are likely to cause both short-term reactions, and long-term illnesses such as certain types of cancers.
Types of Cancers Likely Caused by CPAP and BiPAP Devices
CPAP and BiLevel PAP customers are found to be risk for developing cancer in the following organs:
- Lung
- Liver
- Kidney
- Stomach
Short-Term Health Risks from CPAP and BiPAP Devices
Users of these devices may be at risk for developing the following symptoms of illness:
- Asthma
- Irritation of the skin, eyes, and respiratory tract
- Coughing and shortness and breath
- Chronic inflammation
- Allergic reactions
- Common to severe headaches
- Harmful effects to organs such as kidneys and liver
- Nausea, vomiting, and diarrhea
Research into how and why these devices may cause illnesses is ongoing. More findings are expected to be presented in the coming weeks.
How Philips CPAP & BiPAP Devices Raise Cancer Risk
The sound abatement foam used in their CPAP and BiLevel PAP devices, used to reduce the amount of noise these machines make, may emit cancer-causing particles when it deteriorates, which users could inhale.
Conditions That Cause Foam Degradation in Positive Airway Pressure Devices
Philips has found the following to be the greatest contributing factors to foam degradation:
- Storing devices in places with high temperatures and humidity
- Waiting longer than 5 years to replace a BiLevel PAP or CPAP device
- Using unapproved cleaning methods such as ozone
Research is ongoing to determine all conditions that accelerate the breakdown of the foam material. It is possible that degradation may be caused by more conditions than these listed, some of which may not be in the user’s control.
All Recalled US Philips Respironics Sleep and Respiratory Care Devices
Users of the following CPAP and BiLevel CPAP devices are advised by Philips to stop treatment with these machines immediately, and consult with their physicians before continuing therapy.
Recalled CPAP & BiLevel PAP Devices
- DreamStation ASV (Also known as DreamStation BiPAP autoSV
- DreamStation ST, AVAPS
- DreamStation GO CPAP, APAP, Auto CPAP
- DreamStation CPAP, Auto CPAP, BiPAP
- SystemOne ASV4
- System One 50 Series
- SystemOne 60 Series
- C Series S/T, AVAPS
- OmniLab Advanced Plus
Recalled Mechanical Ventilators
Those using the following ventilators are advised by Philips to continue any life-sustaining ventilation therapy prescribed by their physician.
- Trilogy 100 Ventilator
- Trilogy 200 Ventilator
- A-Series BiPAP V30 Auto Ventilator
Consult with a doctor as soon as possible to discuss the next stages of treatment.
Consult with a Philips CPAP & BiPAP Toxic Tort Lawyer
Users of CPAP and BiPAP machines who are suffering from one or more of the symptoms or illnesses above are advised to seek a free consultation with a toxic tort lawyer as soon as possible to explore their legal options, and discuss the value of their case.
For over 50 years, the toxic tort lawyers at Sobo & Sobo have helped victims across New York, New Jersey and Chicago, IL win settlements that help restore their quality of life, and regain financial and emotional wellbeing in trying times. Contact us online to book your consultation, or call 855-468-7626 to get started today.